Please click on the tables and figures to enlarge
Paediatric sedation in dentistry: an audit of safety, side-effects and patient satisfaction
C. Zhang1 BSc (Xiamen), MSc (King’s College London), PhD (King’s College London)
W J. Botha2 MD (Pretoria), PGDip Sedation (Stellenbosch)
N. Sturzenbaum*3 Zahnärztin (Würzburg), Dr med. dent. (Jena)
1Research Scientist, School of Cancer & Pharmaceutical Sciences, King’s College London, London, SE1 7NH
2Sedationist, Sedation Solutions, 1 Oakcroft Rd, Chessington, Surbiton KT9 1BD
3Director and Principal Dentist, Toothbeary Practice Richmond, 358A Richmond Rd, East Twickenham, TW1 2DU
*Correspondence to: Dr Nicole Sturzenbaum
Email: nicole@toothbeary.co.uk
DOI: 10.63507/QUJF4135
Abstract
Sedation in paediatric dentistry is essential for managing anxiety, pain and cooperation challenges, but it requires adherence to rigorous safety protocols to minimise risks. This audit evaluated the safety, efficacy and patient experience of intravenous (IV) sedation in paediatric dental procedures at a primary care dental practice between January 2023 and September 2024. A total of 1,000 sedation procedures were analysed, assessing patient demographics, sedation drugs (ketamine, propofol), physiological responses, adverse events and parental satisfaction. IV sedation was provided by a medically qualified and experienced practitioner with specific training in the techniques used, and with former anaesthetic experience providing crucial airway management knowledge and skills and supported by sedation-trained dentist and nurses. The facilities for sedation within the primary care setting were inspected and deemed ‘equivalent’ to provide IV sedation in line with current IACSD Standards. Key findings include the absence of serious adverse events or escalation of care, a low incidence of oxygen desaturation events (3%), minimal recovery time variation across ASA I and II classifications and high parental satisfaction (99.9%). Minor adverse events, such as vomiting, were reported being more common in younger patients receiving higher doses of ketamine. The audit suggests that IV sedation is generally safe with a favourable patient and parental experience. However, it highlights the need for refined dosing protocols to mitigate vomiting risks and improve monitoring methods.
Key learning points
- IV sedation in primary care paediatric dentistry, by a trained and experienced non-anaesthetist, when following established protocols, is generally safe with minimal adverse events and high parental satisfaction
- Ketamine use in sedation requires careful dosing to minimise side effects like vomiting, particularly in younger children, emphasising the need for refined dosing strategies
- Further study is needed to better understand the degree of potential adverse events and any potential correlation with other patient parameters.
Introduction
Sedation in paediatric dentistry plays a crucial role in providing safe, effective care to young patients who may experience anxiety, pain or difficulty cooperating during dental procedures.1 However, with the benefits of sedation come inherent risks, underscoring the importance of adherence to established safety guidelines and continuous monitoring of sedation outcomes.2 In the UK, the Intercollegiate Advisory Committee for Sedation in Dentistry (IACSD) provides essential guidelines that help ensure the safe and effective use of sedative techniques in dental settings. These guidelines outline the necessary training, equipment, monitoring and patient assessment criteria for sedation, serving as a cornerstone for best practices within the field.3
There is currently a notable lack of high quality, peer reviewed evidence relating to dental conscious sedation techniques, especially in regards to the safety and efficacy of novel sedation drugs (and combinations of drugs) which may be suitable for children.4 An audit of a primary care paediatric dental sedation service would aid in the evaluation and assessment of the safety, efficacy and patient experience of sedation which would be further enhanced by drawing upon insights from global studies, notably the findings of the Adverse Event Sedation Reporting Tool (AEsSRT).5,6 These large scale studies of sedation-related adverse events provide valuable data on the frequency and nature of complications across diverse settings, serving as a benchmark for assessing the safety of paediatric dental sedation. The findings from the AEsSRT highlight the critical importance of robust sedation protocols and proactive risk management strategies, reinforcing the need for ongoing audits to improve patient outcomes and minimise adverse incidents.
Here we present an audit which examined patient demographics, sedative drugs used and sedation-related adverse events, with a focus on identifying areas for improvement in service delivery and patient care. By combining local audit data with insights from broader international research,7 the audit aims to contribute to the ongoing conversation about sedation safety in paediatric dentistry, providing actionable recommendations for enhancing practice standards and reducing sedation-related risks.
Methods
Patient cohort
The clinic involved in this audit was granted ‘equivalence’ status under IACSD in 2021, allowing the use of IV sedation for children under the age of 12. This includes both the knowledge and skills of the dentists and sedationist involved, as well as the facilities within the practice.3
Children attending the clinic, or who were referred for specific treatment from another practice, were all pre-assessed to determine the extent of treatment required, appropriate means of delivering treatment (different options ranging from local anaesthesia and behaviour management techniques only, or in combination with nitrous oxide or intravenous sedation) and to obtain a comprehensive dental and medical history. Only children who were classified as an American Society of Anaesthesiologists (ASA) 1 or 2 were considered for intravenous sedation.8 None of the children within the audit had received sedation or general anaesthesia for dental treatment before.
Parents and children received a detailed explanation of the sedation options and procedures before deciding on how to proceed.
Intravenous sedation technique
- All sedation cases were conducted within a primary care dental facility by an experienced, medically trained sedationist (non- anaesthesiologist)9,10
- All children fasted in accordance with international standards for general anaesthesia (solid food >6hours, clear liquids >2hours)11
- Successful IV access (with IV cannula) was obtained by using a combination of topical anaesthesia, child-friendly approach and distraction techniques. No pre-medication was used and limited restraint with parental consent and assistance was only used as a last resort
- Parents were encouraged to participate during cannulation (to comfort, support and assist in distraction techniques) and after initial bolus dose of sedative was administered, to stay present until all monitoring equipment was in place and adequate sedation had been achieved. At this point, parents were instructed to leave the treatment area to allow complete focus from the sedation team and minimise distraction. (Parents were invited back as soon as the patient was transferred to the post- sedation recovery area)
- Following secured IV access, all children received a bolus dose of ketamine (0.2-0.5 mg/kg) followed by propofol as a target-controlled infusion (TCI). Propofol TCI were delivered using Paedfusor or Kataria models, with a starting plasma concentration target of 2.0 mcg/ml (children under 30 kg body weight), 1.5 mcg/ml (30-50 kg) or 1 mcg/ml (over 50 kg). For overweight or obese children the ideal body weight was used for the TCI parameters
- Propofol TCI levels were then titrated to achieve the optimal level of sedation to enable treatment and to maintain the appropriate level of consciousness and protective airway reflexes
- All patients were closely monitored using a pulse oximetry, ECG, capnography (end tidal CO2) and received additional oxygen via nasal cannula at 1 l/min
- Dental treatment was carried out, where possible, using a barrier device (rubber dam) to prevent water causing coughing
- On completion of treatment, and when deemed ready, patients were transferred to a private recovery room where they were nursed in the recovery position with continuous pulse oximetry under direct care of a registered general nurse experienced in paediatric anaesthetic care until fully alert and recovered
- Once full discharge criteria were met and all post-sedation information and advice had been provided to the patient and carers, they were discharged
- A follow up phone call was arranged for the day after their treatment, as well as another clinical review approximately two weeks after treatment.
Audit
The audit analysed paediatric patients who received intravenous (IV) sedation for dental treatment at Toothbeary paediatric dental practice between January 2023 and September 2024. Eligible patients were identified through dental records stored in the Carestream R4+® (Carestream Dental, Atlanta, USA) system.
Data from the sedation records of 1,000 patients were collected retrospectively and analysed anonymously. Variables included:
- Patient details: gender, age, weight and ASA classification at preassessment
- Treatment details: type of dental procedure performed, procedure duration and oxygen saturation levels during sedation
- Sedation details: duration of sedation, doses of midazolam, ketamine and propofol administered and recovery time within the clinic
- Post-sedation outcomes: incidences of adverse events such as pain, vomiting or behavioural changes observed during a 24-hour follow-up, along with parental satisfaction ratings.
The extracted data were input into RStudio (version 2024.09.0+375) for statistical analysis.
Results
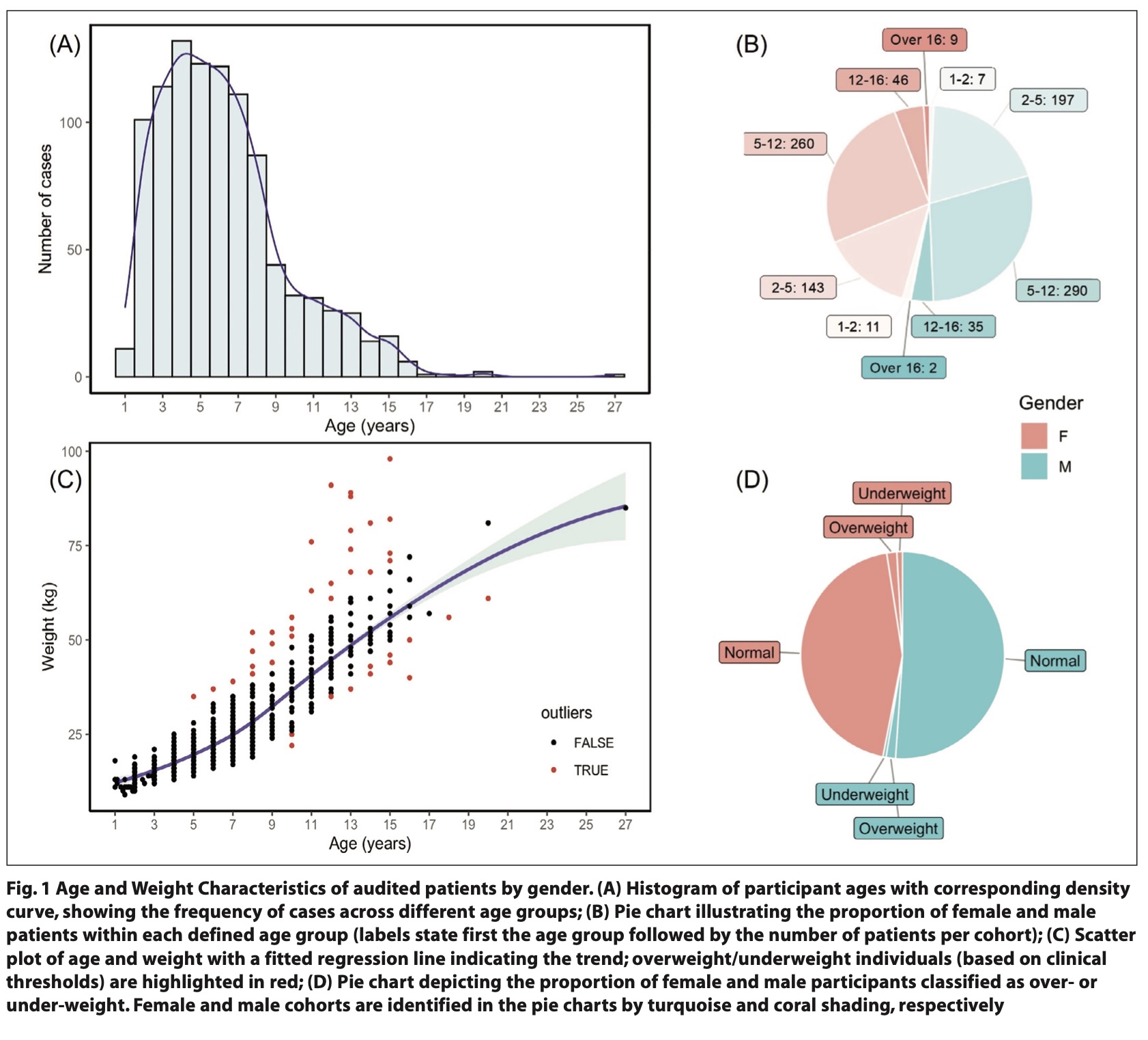
This audit examined 1,000 sedation procedures performed on paediatric patients between January 2023 and September 2024, of which 469 were female and 531 were male. No patients received or required a repeat sedation session for previous or follow-up dental treatment at Toothbeary during the period covered by the audit. However, the authors do not know if the patients required repeat procedures at other providers as the patients were not followed up.
The majority of children receiving sedation were in the age groups of two to five years and five to twelve years (Fig. 1A), with no significant gender differences across these age categories (Fig. 1B). This finding highlights the need for sedation in this younger age group, which under current guidelines are mostly restricted to secondary care facilities.3 The demographic and physical characteristics of patients, such as weight distribution and associated health factors, were considered carefully in interpreting sedation outcomes.
Of the total cohort, abnormal weight classifications were observed primarily in the older paediatric group (ages 12 - 16) (Fig. 1C) with no marked differences between the male and female cohorts (Fig. 1D). In comparison, weight irregularities were less common in younger age groups, suggesting that the 12 – 16 year group may require special consideration regarding weight when considering IV sedation as a treatment option.
Oxygen desaturation events
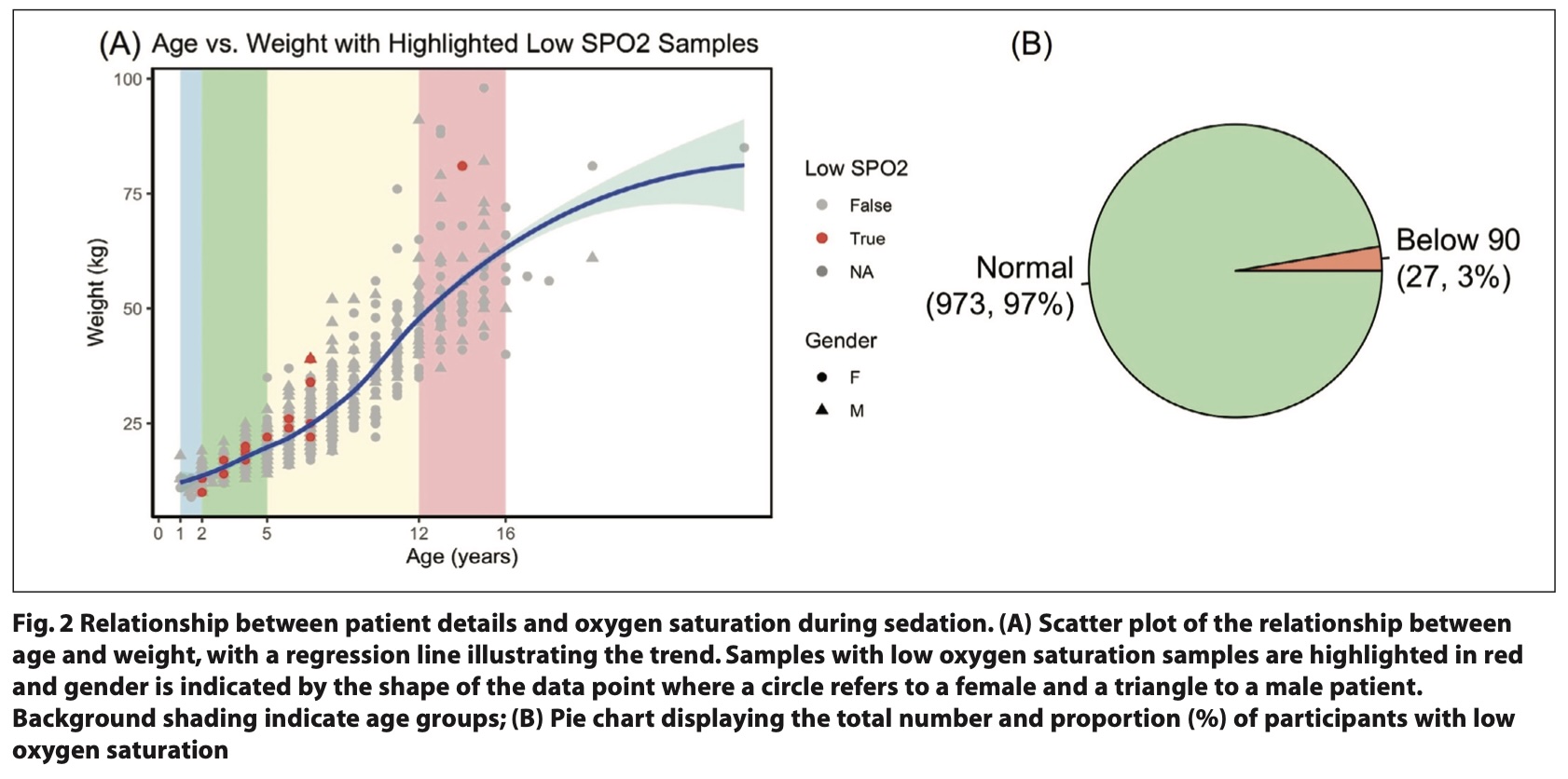
In terms of physiological responses during sedation, heart rate and oxygen saturation levels were closely monitored. Values were obtained from electronic monitoring during sedation, with oxygen saturation and pulse readings digitally recorded at five-minute intervals (Fig. 2A).
An overall low incidence of desaturation events was noted: only 3% of patients had oxygen saturation levels drop below 90% during sedation, showing that desaturation was relatively rare in this cohort (Fig. 2B). The ratio of males to females experiencing oxygen levels below 90% was nearly equal, with 14 boys and 13 girls showing this decline (Fig. 1A).
No correlation was observed between desaturation events and other parameters including ASA status, duration of treatment, dose of ketamine and propofol infusion rates (data not shown).
Treatment and recovery duration
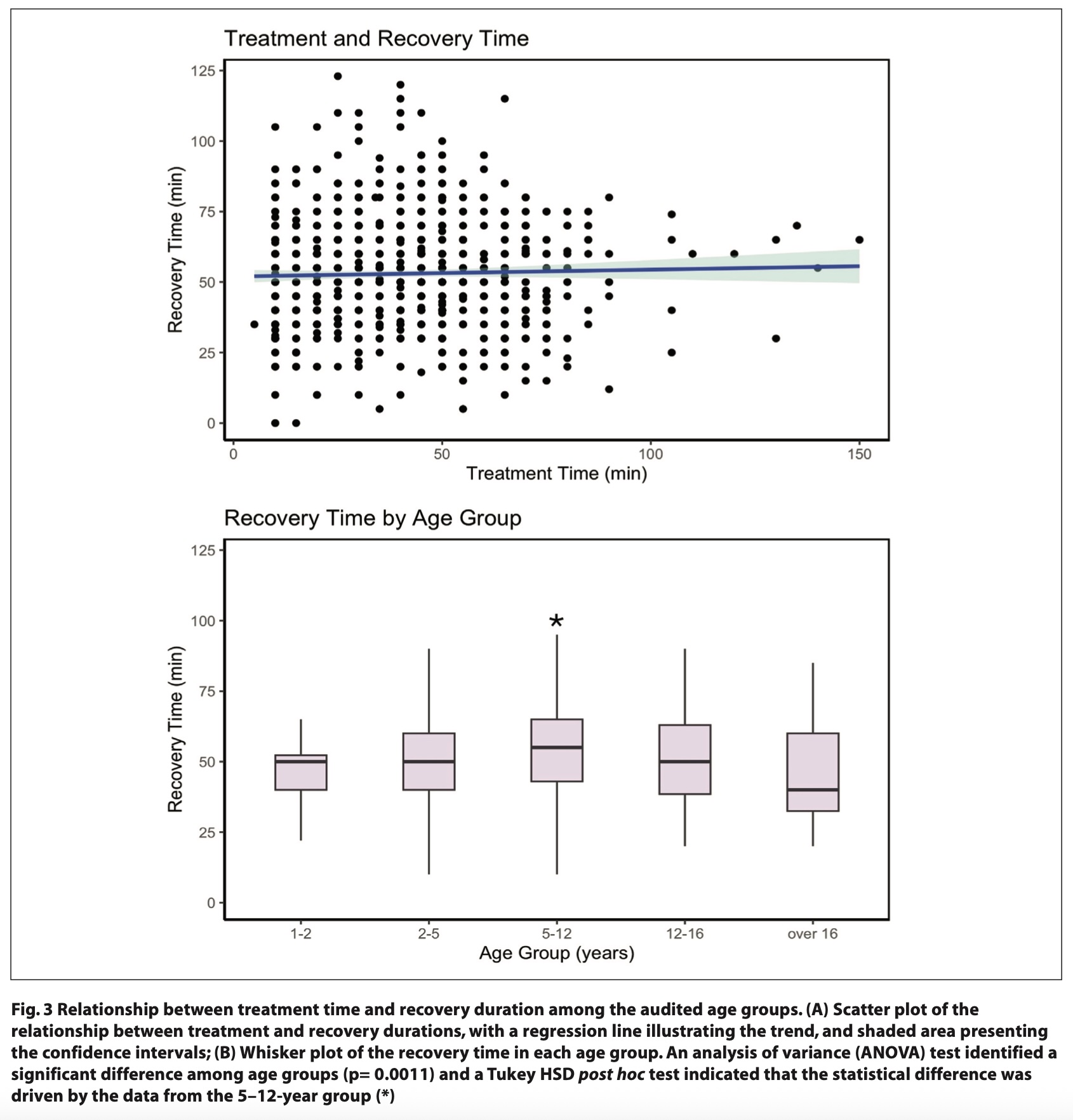
The duration of dental procedures varied widely, from a minimum of 5 minutes to a maximum of 150 minutes, highlighting the variability in treatment complexity and patient needs. Post- sedation recovery times in the clinic ranged from 0 to 123 minutes, with notable variation between different age groups. Statistical analysis indicated no significant correlation between treatment duration and recovery time (Fig. 3A). This suggests that other factors beyond the length of the procedure, such as age or individual response to sedative agents, may influence recovery duration.
The recovery time data revealed a statistically significant difference across age groups, with the most prolonged recovery times recorded for children in the 5 – 12 year group, where the average recovery time was 55.1 minutes (Fig. 3B). A shorter mean recovery time of 44.4 minutes was observed in the youngest children (ages 1 - 2), although the smaller sample size for this group (18 cases, representing 1.8% of the total cohort) limited the statistical power of these findings in post-hoc tests. The age-related differences in recovery time may reflect developmental variations in metabolism and tolerance to sedative agents and suggest that age-specific guidelines could enhance post-sedation management.
When analysing recovery times by ASA classification, children classified as ASA 1 (810 cases) and ASA 2 (190 cases) exhibited comparable mean and median recovery times, indicating that general health status, within this sample’s ASA 1 and 2 classifications, did not substantially impact recovery durations. This finding supports the current ASA-based stratification for determining sedation suitability in a primary care setting and suggests that ASA classifications may be a reliable indicator of recovery time expectations in ASA 1 and 2 paediatric patients.
Post-sedation adverse events and parent satisfaction
A post-sedation follow-up was conducted the day after treatment, where parents were phoned to enquire about encountered side effects and their overall satisfaction with the sedation process and dental treatment outcome. Overall, satisfaction was high, with 99.9% of parents reporting that they were either ‘very satisfied’ or ‘satisfied' with the outcome. However, despite the high satisfaction rates, some minor adverse events were reported. Specifically, 32% of cases (319 patients) reported post-treatment pain, fever or minor cosmetic concerns. Pain was the most frequently reported side effect, typically subsiding within two days post-procedure. Only one parent expressed dissatisfaction, citing a perceived lack of information about the sedation process. This high satisfaction rate suggests that, with good communication and comprehensive monitoring, IV sedation in paediatric dental care is both safe and valued by families.
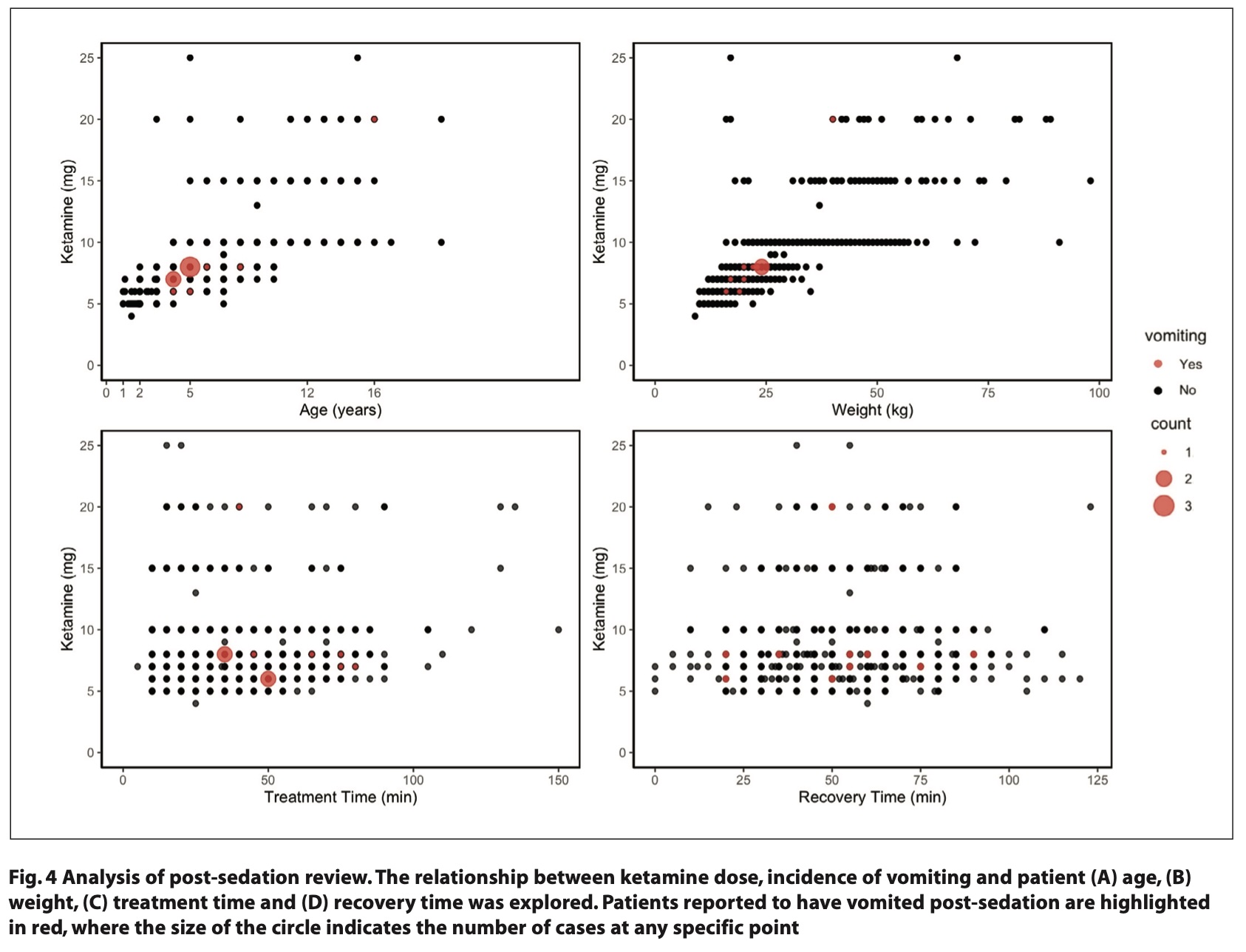
Among adverse events, vomiting was recorded in 11 patients (1.1%) within the first 24 hours following sedation. Of these cases, ten patients had received IV ketamine, in line with the known association between ketamine and postsedation emesis.12 Patient and treatment details for these cases are presented in Figure 4 separated by age (Fig. 4A), weight (Fig. 4B), treatment time (Fig. 4C) and recovery time (Fig. 4D). The majority of vomiting cases were clustered among younger children, with four cases involving 5-year-olds and three involving 4-year-olds. Five of these cases had been administered an 8 mg dose of titrated ketamine, while the other cases received slightly lower doses (6 or 7 mg). Only one child received oral ketamine (250 mg; 4 mg/kg) and subsequently experienced vomiting. This pattern suggests a possible dose response relationship between ketamine and vomiting incidence, which could inform future protocols for dose adjustments in younger patients.
Discussion
This audit demonstrates a generally safe and well received application of IV sedation in paediatric dental patients in a primary care facility, with low incidences of adverse events and a total absence of severe complications. It is important to note that the training and experience of the sedationist, in particular, experience in airway management, is central to the safety of this particular sedation technique. Equally, working within a consistent and well trained and acquainted team is as important as the skills and experience of the person leading the sedation service.
The absence of repeat sedations demonstrates that the dental treatment provided was both appropriate and effective. Variations in recovery times between age groups were noted, particularly for the 5 to 12 year age group, suggesting that age may influence recovery duration following sedation. While minor adverse effects such as pain and vomiting were recorded, they were managed effectively within the clinical setting. High parental satisfaction and minimal adverse feedback affirm the efficacy and safety of the sedation protocols used. Further research could explore optimised dosing strategies for reducing the incidence of vomiting associated with ketamine in young patients, as well as protocols for managing paediatric patients with atypical weight classifications to ensure safer sedation outcomes.
The ability to successfully cannulate, sedate and complete treatment in all cases within this audit is a testimony to the use of having a well-planned and structured patient journey, using well trained staff with a team approach and using a sedation technique that allows fine titration and flexibility to accommodate the considerable inter-patient variability in response to sedatives typically seen within this patient cohort.
The authors acknowledge the omission of blood pressure monitoring as part of the sedation protocol for young children. This was done to limit the agitation caused by blood pressure monitoring when compared to the clinical significance of any changes in blood pressure under IV sedation in this patient cohort. Many studies of the use of propofol for sedation in children have shown that blood pressure changes during sedation are brief, transient and well tolerated.13,14 On balance, it was decided rather to use clinical judgement and patient symptoms as a guide to adequate blood pressure levels before discharge, as children found blood pressure monitoring too uncomfortable and upsetting to justify its routine use. As part of the discharge criteria, all patients were required to be free of any signs or symptoms that might suggest persistent hypotension (light headedness, dizziness when ambulatory, or looking pale, sweaty or unwell). For any future prospective studies, including blood pressure monitoring will help to confirm this view and further justify this approach.
Additionally, objective monitoring of oxygen desaturation events in this environment is challenging, the measurement at five-minute intervals has one caveat: although being objective, it can only provide a snapshot of conditions at that moment in time and, crucially, does not indicate the true nature, severity and duration of desaturation. Significant artefact also occurs frequently during treatment under sedation, most often due to patient movement and dislodging of the oxygen saturation probe, leading to incorrect or false readings.
In conclusion, this audit found that all patients were treated successfully and discharged home following their treatment under sedation with very low levels of sedation-related side effects and very high patient satisfaction scores. No repeat sedation sessions were required / performed at Toothbeary. However, given that no long-term follow-up was performed, it is not possible to determine whether patients received repeat procedures at other providers.
For future studies, the following areas would prove useful for further investigation:
- A more in-depth look at adverse events (nature, severity, duration)
- Establishing a more reliable and, preferably objective, method of data capture
- Further investigation of any link between adverse events and ASA status, length of treatment and dose / combination of sedatives used
- Widening of the Patient Reported Outcome Measures (PROMS) and Patient Reported Experience Measures (PREMS) to provide a more detailed and holistic picture of the quality of care provided.
Acknowledgements
We thank the Toothbeary team for the meticulous logging and collection of patient data. In addition, we acknowledge Prof Stephen Sturzenbaum’s advice pertaining to the experimental analysis and invaluable guidance on the refinement of the paper.
Declaration of Interests
The authors declare no conflicts of interest.
Ethics
The nature of this audit required no ethical committee approval.
References
1. Hosey M T. Managing anxious children: The use of conscious sedation in paediatric dentistry. Int J Paediatr Dent 2002; 12: 359–372. DOI: 10.1046/j.1365-263X.2002.00389.x.
2. Coté C J, Wilson S. Guidelines for monitoring and management of pediatric patients before, during, and after sedation for diagnostic and therapeutic procedures. Pediatrics 2019; 143: e20191000. DOI: 10.1542/peds.2019-1000.
3. Intercollegiate Advisory Committee for Sedation in Dentistry (IACSD). IACSD Guidelines on Sedation Safety in Dentistry. Standards for Conscious Sedation in the Provision of Dental Care. Faculty of Dental Surgery, Royal College of Surgeons of England, 2020. https://www.saad.org.uk/IACSD%202020.pdf (accessed December 2024).
4. Ashley P F, Chaudhary M, Lourenço-Matharu L. Sedation of children undergoing dental treatment. Cochrane Database Syst Rev 2018; 12: CD003877. DOI: 10.1002/14651858.CD003877.pub5.
5. Adverse event reporting tool to standardize the reporting and tracking of adverse events during procedural sedation: a consensus document from the world SIVA international sedation task force. Br J Anaesth 2012; 108:13-20. DOI: 10.1093/bja/aer407.
6. Mason K P, Roback M G, Chrisp D, et al. Results from the adverse event sedation reporting tool: a global anthology of 7952 records derived from >160,000 procedural sedation encounters. J Clin Med 2019; 8: 2087. DOI: 10.3390/jcm8122087.
7. Williams M R, Nayshtut M, Hoefnagel A, et al. Efficacy outcome measures for pediatric procedural sedation clinical trials: an ACTTION systematic review. Anesth Analg 2018; 126: 956-967. DOI: 10.1213/ANE.0000000000002456.
8. Statement on ASA physical status classification system. 2020. Online information available at https://www.asahq.org/standards-andpractice-parameters/statement-on-asa-physical-status-classificationsystem (accessed December 2024).
9. Academy of Medical Royal Colleges. (2013). Safe sedation practice for healthcare procedures: standards and guidance. Academy of Medical Royal Colleges, London. Online information available at https://www.aomrc.org.uk/wpcontent/uploads/2024/05/Safe_Sedation_Practice_1213.pdf (accessed December 2024).
10. Academy of Medical Royal Colleges. (2021). Safe sedation practice for healthcare procedures: an update. Academy of Medical Royal Colleges, London. Online information available at https://www.aomrc.org.uk/wpcontent/uploads/2021/02/Safe_sedation_practice_for_healthcare_procedures_update_0521.pdf (accessed December 2024).
11. Arun B G, Korula G. Preoperative fasting in children: an audit and its implications in a tertiary care hospital. J Anaesthesiol Clin Pharmacol 2013; 29: 88-91. DOI: 10.4103/0970-9185.105810.
12. Green S M, Roback M G, Kennedy R M, Krauss B. Clinical practice guideline for emergency department ketamine dissociative sedation: 2011 update. Ann Emerg Med 2009; 57: 449–461. DOI: 10.1016/j.annemergmed.2010.11.030.
13. Bassett, Kathlene E et al. Propofol for procedural sedation in children in the emergency department. Annals of Emergency Medicine, Volume 42, Issue 6, 773 –782. DOI: 10.1016/s0196-0644(03)00619-x.
14. Guenther, Elisabeth et al. Propofol sedation by emergency physicians for elective pediatric outpatient procedures. Annals of Emergency Medicine, Volume 42, Issue 6, 783 – 791. DOI: 10.1016/s0196-0644(03)00634-6.