Please click on the tables and figures to enlarge
Peri-operative management of dental patients requiring procedural sedation and taking Ozempic®, Mounjaro®, or other glucagon-like peptide-1 receptor agonist- containing medications
I. Khan*1, 2 BDS, Cert (Con Sed) (UoP), MFDS, DSCD RCSEd
1Senior Dental Surgeon (Special Care Dentistry), Midlands Partnership University NHS Foundation Trust - Dental Services, Cannock Hospital Dental Department, Cannock, Brunswick Road, WS11 2XY
2Honorary Assistant Professor, College of Medicine and Health, School of Dentistry, University of Birmingham, Birmingham, B15 2TT
*Correspondence to: Mr Ishfaq Khan
Email: dental.research@mpft.nhs.uk
DOI: 10.63507/OODO3230
Abstract
The rising number of prescriptions for glucagon-like peptide-1 receptor agonists and glucose-dependent insulinotropic polypeptide receptor agonists is due to expanding indications and increased demand. These medications can cause delayed gastric emptying and increase the risk of pulmonary aspiration, particularly during deep sedation and general anaesthesia. Special care patients, such as those with Parkinson's disease or cerebral palsy, are at higher risk. This paper examines the pharmacology of glucagon-like peptide-1 receptor agonists and glucose-dependent insulinotropic polypeptide receptor agonists, assesses the risk of pulmonary aspiration under procedural sedation, reviews current guidance from anaesthetic societies and suggests methods to mitigate risks for patients on these medications during procedural sedation.
Key learning points
- Increase awareness of glucagon-like peptide-1 receptor agonists and glucose-dependent insulinotropic polypeptide receptor agonists licensed in the UK and their significance for sedationists
- Outline current guidance on the peri-operative management of patients taking glucagon-like peptide-1 receptor agonists and glucose-dependent insulinotropic polypeptide receptor agonists for procedural sedation and general anaesthesia
- Discuss methods to reduce the risk of pulmonary aspiration during procedural sedation.
Introduction
Pharmacological evaluation is crucial during pre-assessment for dental patients needing sedation. Certain medications can affect benzodiazepine metabolism, or patients taking fall risk-increasing drugs (FRIDs) may have a higher risk of falls post-sedation.1
Another group of medicines that is becoming increasingly important for procedural sedation and general anaesthesia include glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and glucose-dependent insulinotropic polypeptide receptor agonists (GIP RAs).
GLP-1 RAs and GIP RAs can be associated with delayed gastric emptying and pulmonary aspiration despite recommended pre-operative fasting during deep sedation or general anaesthesia.2, 3 The Food and Drug Administration (FDA) Center for Drug Evaluation and Research updated its drug packaging for GLP- 1 RAs to warn patients and healthcare providers of this risk,4 with the European Medicines Agency (EMA),5 and Medicines and Healthcare Products Regulatory Agency (MHRA),6 subsequently issuing similar warnings.
The rising demand for these medications for cosmetic weight loss, driven by online pharmacy availability, without physician involvement, has led to significant concerns. Updated guidance from the General Pharmaceutical Council (GPhC) expects pharmacists to now conduct thorough assessments and to actively collaborate with the patient's general medical practitioner before prescribing weight loss medications.7 Patients must also be informed of the risks, including pulmonary aspiration, associated with GLP-1 and GIP RAs.
Pharmacology of GLP-1 RAs and GIP RAs
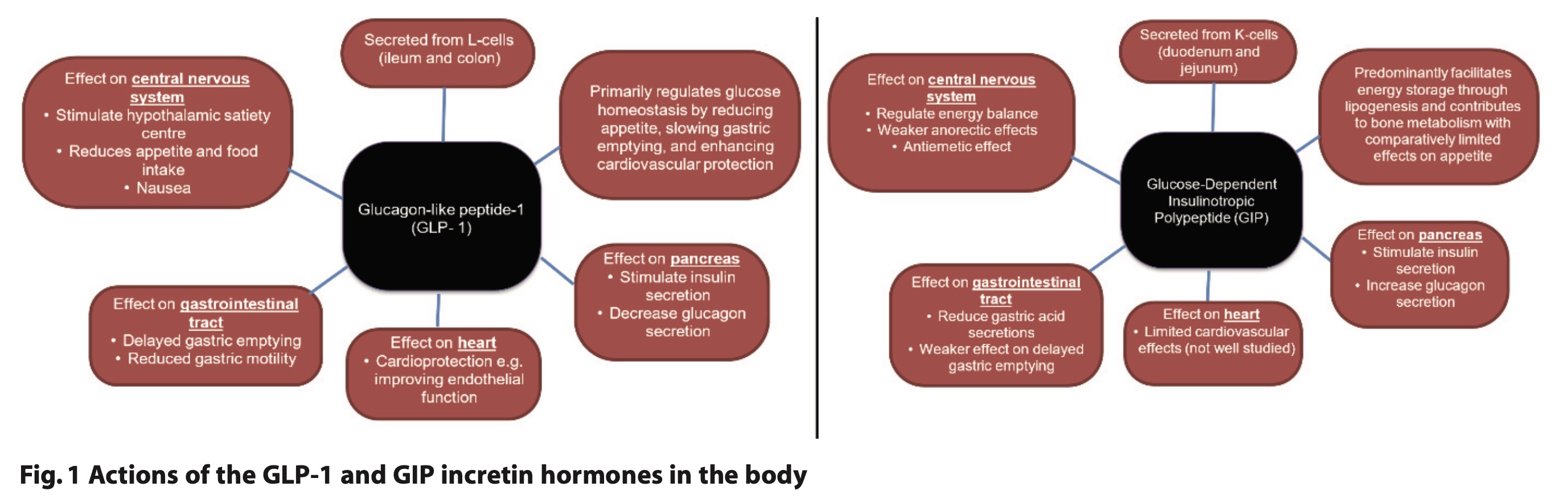
GLP-1 and GIP are incretin hormones released by the body in response to glucose.8 GLP-1 and GIP receptors may be found in the pancreas, central nervous system (CNS), heart and gastrointestinal tract, in addition GLP-1 receptors have been identified in the liver and kidneys (Figure 1).
Activation of GLP-1 and GIP receptors in the pancreas increases insulin secretion from β cells in the islets of Langerhans and GLP-1 RAs inhibit glucagon in pancreatic α-cells, leading to a reduction in blood glucose levels.9
In the CNS, activation decreases appetite and increases satiety.10 Other effects include cardioprotective benefits, such as reduced systolic blood pressure, weight loss, and improved endothelial function.11 In the gastrointestinal system, it leads to reduced gastric motility and gastric secretions and delayed gastric emptying.2
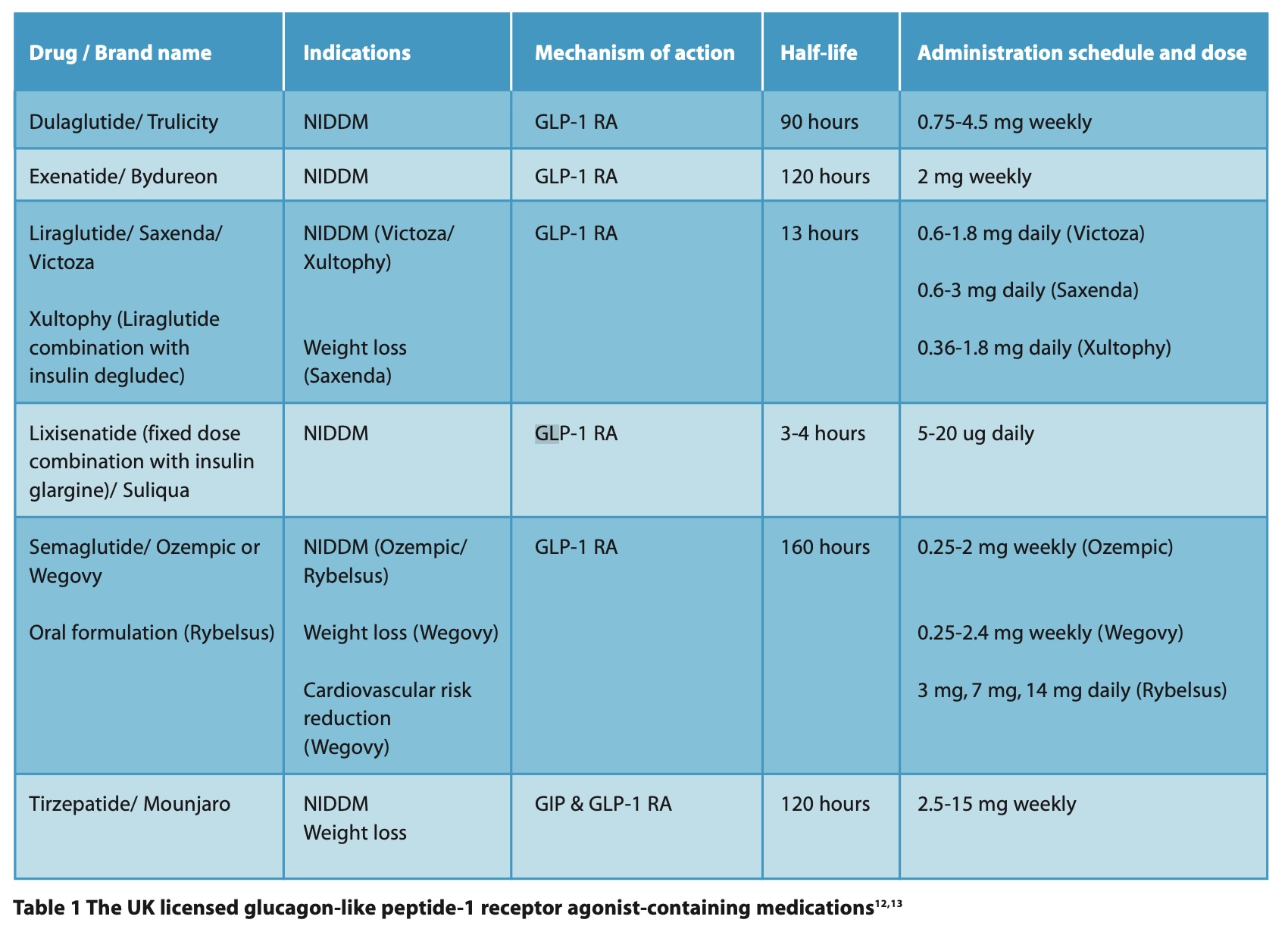
As a result, these medications may, in the long term, help to increase access to dentistry for patients who were previously restricted to bariatric dental services or domiciliary care. GLP-1 and GIP RAs can be recognised by their common suffix ‘-tide’. These medications can be described as short- or long-acting, with patients initially starting at a low dose and gradually increasing it. Routes of administration include oral (daily) or subcutaneous (daily or weekly). They are metabolised by the liver and eliminated by renal excretion.9
They have no interactions with benzodiazepines, nitrous oxide, N-Methyl-D-Aspartate (NMDA) receptor antagonists, or synthetic opioids used in procedural sedation in dentistry.12 They do, however, interact with erythromycin, which can reduce the effectiveness of GLP-1 RAs but also can help to accelerate gastric emptying.12
Five GLP-1 RAs and one dual GIP and GLP-1 RA are licensed in the UK (Table 1).13 GLP-1 and GIP RAs are commonly prescribed for diabetes and weight loss.12 Potential future uses for GLP-1 RAs may include respiratory disease,14 in aesthetic medicine,15 and Alzheimer’s disease.16
Contraindications to GLP-1 RAs include diabetic ketoacidosis, pancreatitis, severe hepatic, renal or gastrointestinal disease12 and patients with medullary thyroid cancer and multiple endocrine neoplasia type 2 due to the potential risk of thyroid c -cell tumours with GLP-1 RAs.2,12
Gastroparesis and pulmonary aspiration
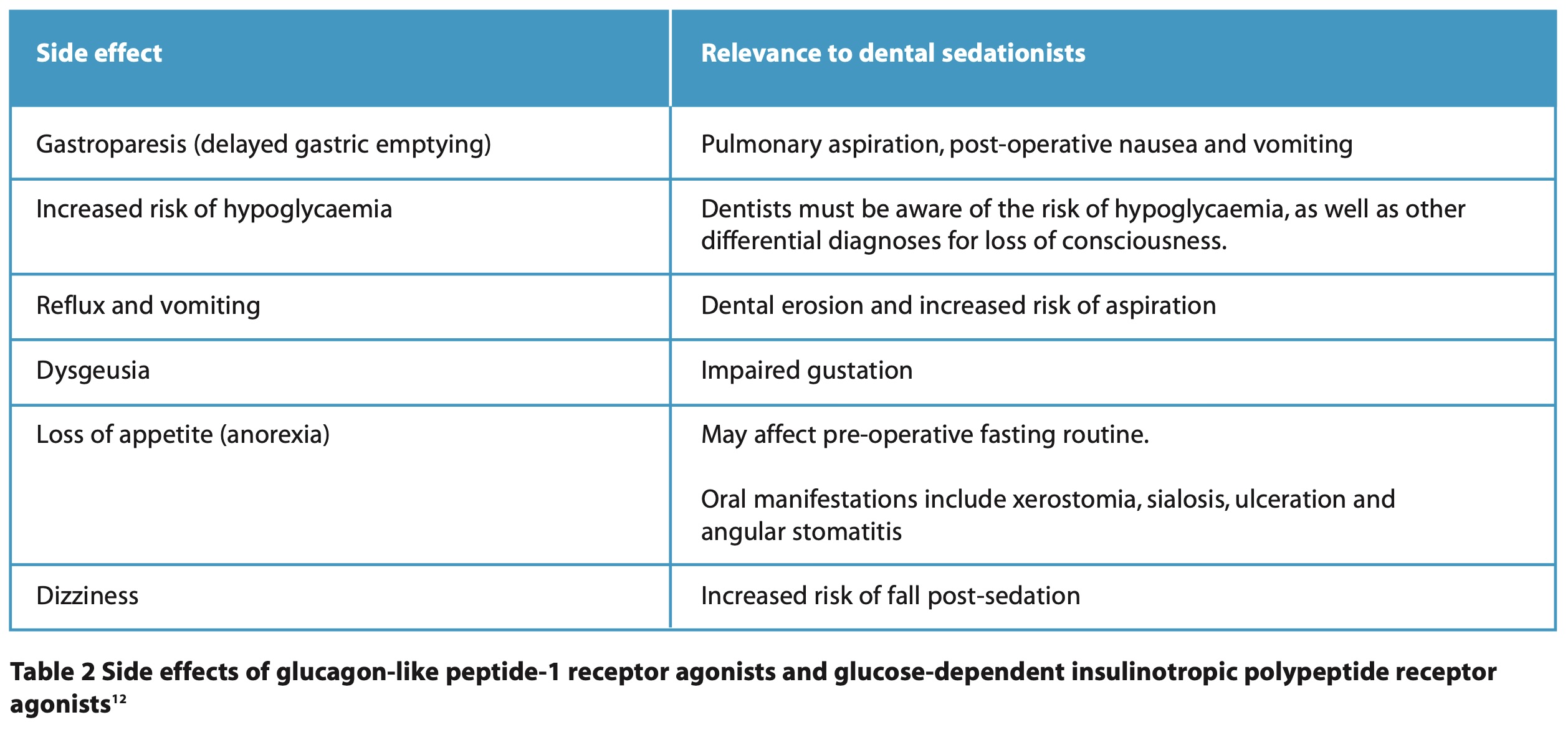
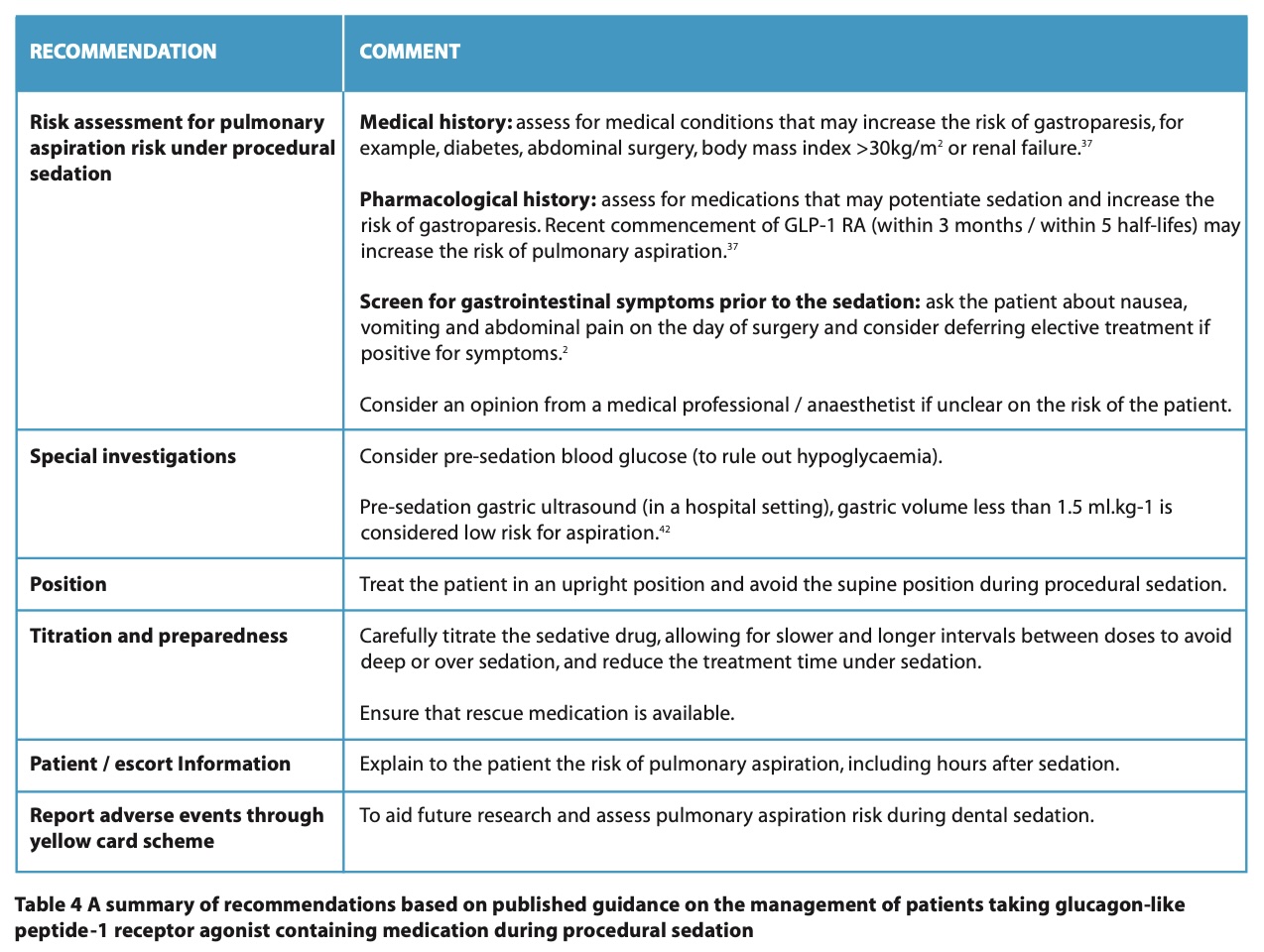
The more common side effects of GLP-1 and GIP RAs of relevance to sedationists are shown in Table 2.12 In this paper we will concentrate on gastroparesis and the risk of pulmonary aspiration with procedural sedation.
There have been several case reports of gastroparesis associated with GLP-1 RAs prior to sedation and general anaesthesia.17-27 Due to this risk, some anaesthetic societies recommend patients stop GLP-1 RA medications and extend peri-operative fasting. Gastroparesis can be associated with older age, diabetes, Parkinson’s disease and postabdominal surgery.28 Gastroparesis may present clinically as chronic early satiety, postprandial fullness, upper abdominal pain, vomiting, nausea and anorexia.28 In addition to GLP-1 and GIP RAs, other drugs associated with gastroparesis include opioids, anticholinergics, alcohol, tobacco and cannabinoids.29 Prevalence of gastroparesis associated with GLP-1 RAs has been reported between ~0.1%,30 and 5%.29
Pulmonary aspiration is the inhalation of oropharyngeal or gastric contents into the larynx and lower respiratory tract, which can lead to aspiration pneumonitis, aspiration pneumonia or, rarely, death.31 Differential diagnoses include acute respiratory distress syndrome, asthma or COPD exacerbation, pulmonary emboli and pulmonary oedema.32 Pulmonary aspiration is associated with deep sedation or general anaesthesia due to loss of protective reflexes, especially prolonged procedures under deep sedation, and if patients are taking other medications that cause gastroparesis.17 It can occur hours after deep sedation or general anaesthesia.
Pulmonary aspiration results in a decrease in alveolar ventilation (patients present with wheeze, cough, rhonchi and rales) apnoea, and cyanosis.32 Additionally, blood pressure falls and patients exhibit tachycardia and tachypnoea.32
Factors which may increase the risk of pulmonary aspiration in patients taking GLP-1 and GIP RAs include patients at the escalation phase of dosing, taking higher doses, longer acting medications, presence of gastrointestinal symptoms of gastroparesis and concomitant evidence of medical conditions associated with gastroparesis.2
Aspiration risk during anaesthesia can be reduced by identifying patients prone to vomiting and reflux, minimising pre-operative gastric contents, reducing emetic stimuli, and preserving protective reflexes.33
Management of pulmonary aspiration in a dental setting includes placing the patient in a head-down tilt or Trendelenburg position (with their head positioned to the right), clearing the airway, providing oxygenation through a face mask or nasal cannula (care should be exercised with bag-valve mask ventilation to avoid gastric insufflation),34 reversal, and, if needed, referral to a hospital setting for observation or if the patient is deteriorating.
Current guidance and recommendations
A systematic review from 2017 which looked at the incidence of pulmonary aspiration during procedural sedation found no cases attributed to dentistry.31 The review found the most common sedative associated with pulmonary aspiration was propofol, followed by benzodiazepines (midazolam and diazepam) and there was also one case attributed to nitrous oxide.31 Ketamine, known for preserving protective airway reflexes, may be a safer alternative for dissociative sedation in patients at higher risk of aspiration.31 The study did not clarify whether any of the patients were taking GLP-1 or GIP RAs.
Since then, there have been at least four cases of gastroparesis associated with GLP-1 RAs and sedation, including during an MRI,17 endoscopic procedures (one patient received a propofol infusion,18 and another received a fentanyl bolus, propofol bolus, and propofol infusion),20 and foot arthrodesis (the patient received block sedation with propofol and ketamine).21 The patient who received a fentanyl bolus, propofol bolus and propofol infusion developed pulmonary aspiration during maintenance; their medical history included reflux, body mass index (BMI) 37 kg/m², and obstructive sleep apnoea.20
Research on the ideal peri-operative management of patients on GLP-1 or GIP RAs is limited, with no consensus among anaesthetic societies. The evidence, mostly from case reports, does not consistently show a significant risk of pulmonary aspiration with GLP-1 RAs.35
The risk of oversedation increases when doses are not titrated and are given as bolus doses, such as with intranasal and oral sedation. Additionally, advanced sedation techniques, including the use of multiple drugs, or propofol may carry a higher risk of oversedation. These techniques are often used for special care patients, who may have a medical history that increases their risk of gastroparesis. Consequently, these patients are at an even higher risk if they are also taking GLP-1 RA-containing medications.
Many patients taking these medications may be obese and already at increased risk of complications from procedural sedation including failed cannulation,36 and have other comorbidities, this needs consideration.
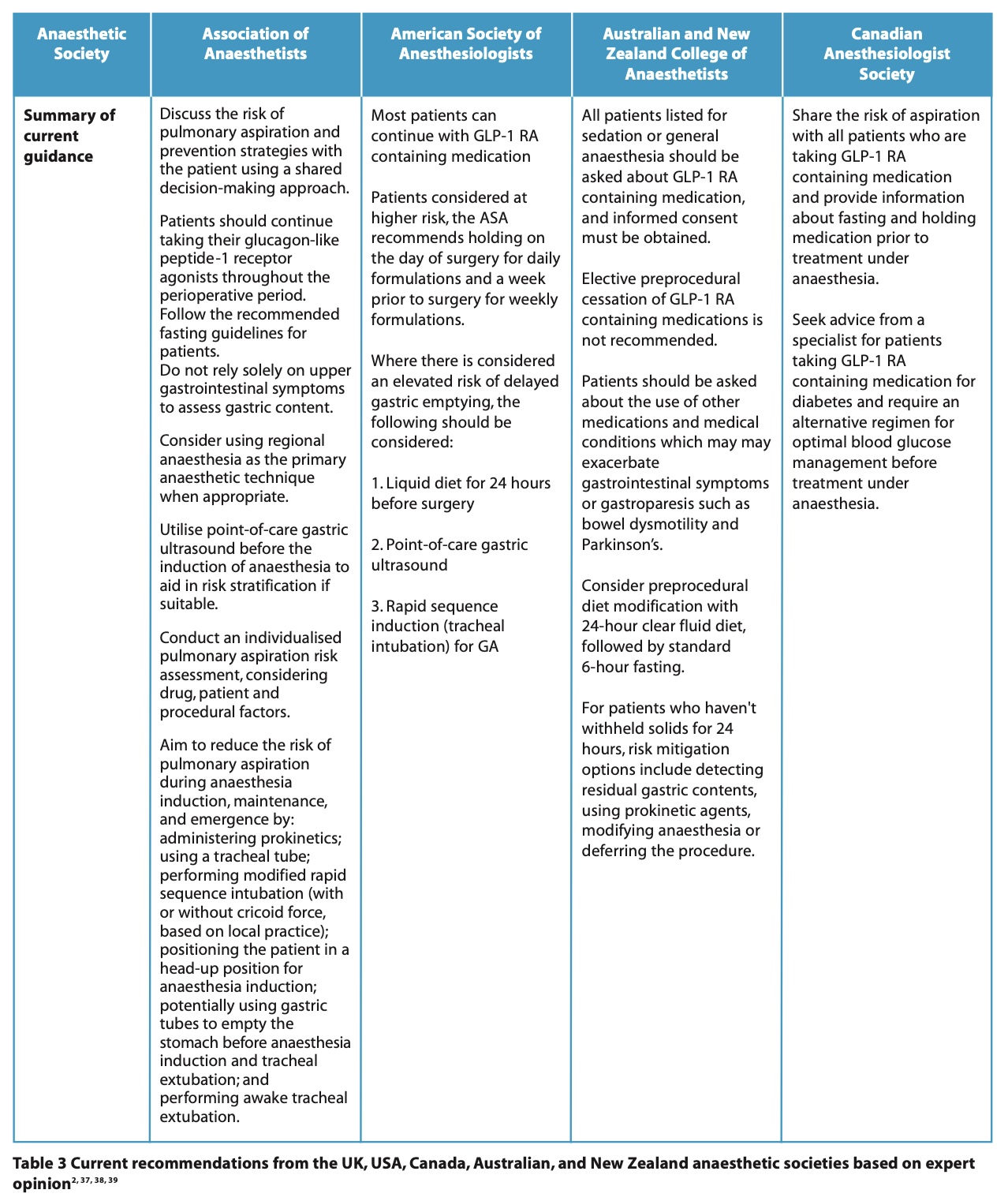
The Association of Anaesthetists,37 American Society of Anesthesiologists (ASA),2 Australian and New Zealand College of Anaesthetists (ANZCA),38 and the Canadian Anesthesiologist Society (CAS),39 have all provided guidance based on expert opinion (Table 3).
The UK currently advises that patients should continue their medication prior to sedation or general anaesthesia, but an individualised approach should be taken to identify patients at higher risk. It is important to note that the UK guidance points out that procedures in which sedation or no tracheal intubation are used may carry a greater risk of pulmonary aspiration.37 Guidance is to be reviewed as more evidence emerges and also as dual and triple agonists are developed.2
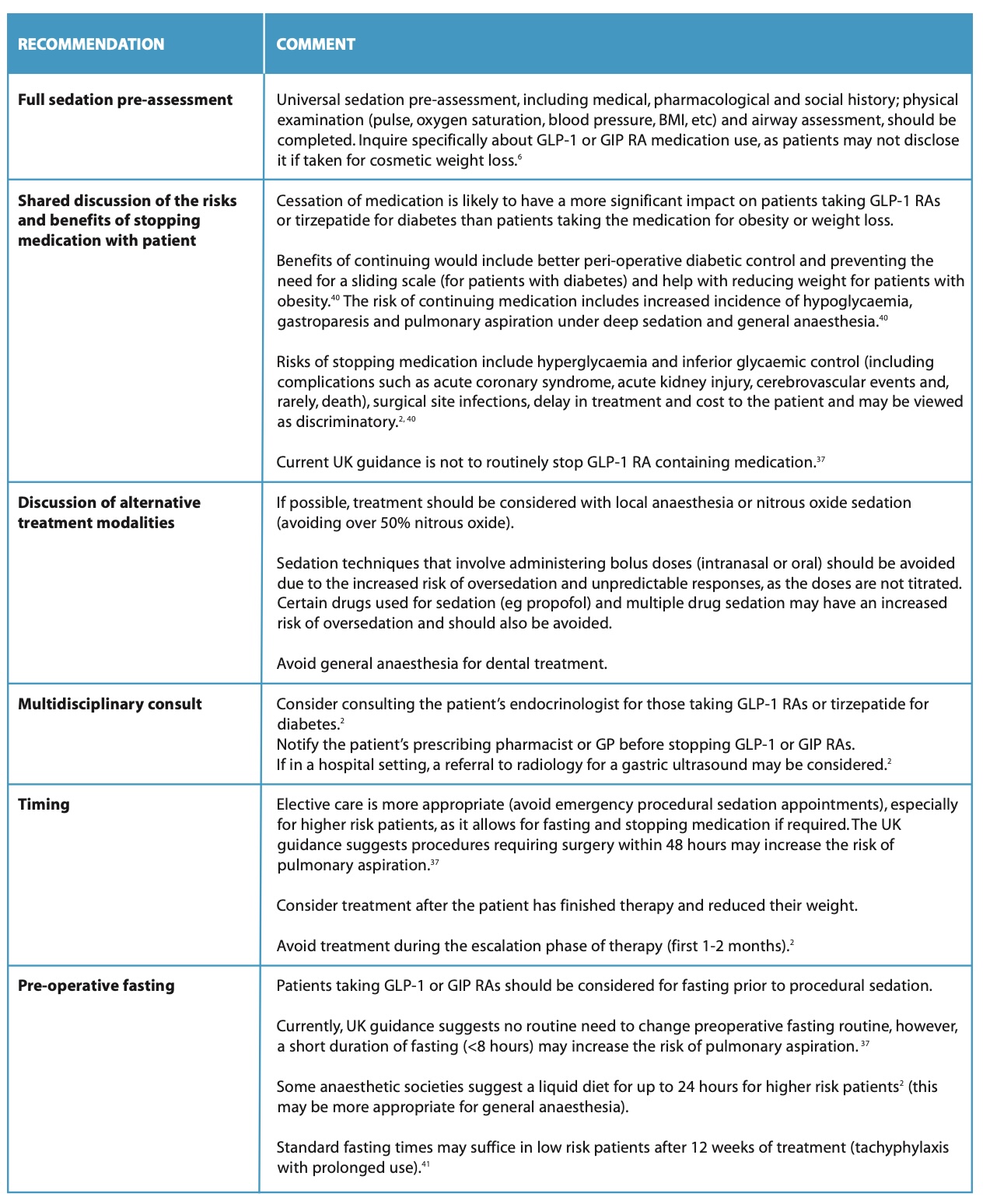
Based on the guidance from the above societies, the following recommendations can be followed by dental sedationists to reduce the risk of pulmonary aspiration in patients receiving GLP-1 or GIP RAs (Table 4).
Conclusion
The rising demand for GLP-1 and GIP RAs means dental sedationists will likely encounter patients on these medications, who may face challenges like pulmonary aspiration during deep sedation. It is important for dental sedationists to follow the latest UK guidelines to prevent complications.
Acknowledgements
The author would like to thank the Research and Library department at Midlands Partnership University (MPFT) NHS Foundation Trust.
Declaration of Interest
None.
References
1. Khan I. Falls: considerations for the dental surgeon. Br Dent J 2020; 228: 509-514. DOI:10.1038/s41415-020-1422-0.
2. Kindel T L, Wang A Y, Wadhwa A et al. Multi-society clinical practice guidance for the safe use of glucagon-like peptide-1 receptor agonists in the perioperative period. Surg Endosc 2024; 6: 180-183. DOI: 10.1007/s00464-024-11263-2.
3. do Nascimento T S, Pereira R O, Maia E et al. The impact of glucagon-like peptide-1 receptor agonists in the patients undergoing anesthesia or sedation: systematic review and meta-analysis. Perioper. Med. 2024; 13: 78. DOI: 10.1186/s13741-024-00439-y.
4. The United States Food and Drug Administration (FDA). Drug Safety-related Labeling Changes (SrLC)- Semaglutide. 2024. Online information available at www.accessdata.fda.gov/scripts/cder/safetylabelingchanges/index.cfm?event=searchdetail.page&DrugNameID=2711 (accessed June 2024).
5. European Medicines Agency. New recommendations for GLP-1 receptor agonists to minimise risk of aspiration and pneumonia aspiration during general anaesthesia or deep sedation. 2024. Online information available at www.ema.europa.eu/en/documents/agenda/agenda-prac-meeting-8-11-july-2024_en.pdf (accessed December 2024).
6. Medicines and Healthcare Products Regulatory Agency (MHRA). GLP-1 and dual GIP/GLP-1 receptor agonists: potential risk of pulmonary aspiration during general anaesthesia or deep sedation. 2025. Online information available at https://www.gov.uk/drug-safetyupdate/glp-1-and-dual-gip-slash-glp-1-receptor-agonists-potential-risk-of-pulmonaryaspiration-during-general-anaesthesia-or-deep-sedation (accessed January 2025).
7. General Pharmaceutical Council (GPhC). Guidance for registered pharmacies providing pharmacy services at a distance, including on the internet. 2025. Online information available: https://assets.pharmacyregulation.org/files/2025-02/gphc-guidance-registeredpharmacies-providing-pharmacy-services-distance-february-2025.pdf (accessed February 2025).
8. Seino Y, Fukushima M, Yabe D. GIP and GLP-1, the two incretin hormones: similarities and differences. J Diabetes Investig 2010; 1: 8-23. DOI: 10.1111/j.2040-1124.2010.00022.x.
9. Sfairopoulos D, Liatis S, Tigas S, Liberopoulos E. Clinical pharmacology of glucagon-like peptide-1 receptor agonists. Hormones. 2018; 17: 333-350. DOI: 10.1007/s42000-018-0038-0.
10. Trapp S, Brierley D I. Brain GLP-1 and the regulation of food intake: GLP-1 action in the brain and its implications for GLP-1 receptor agonists in obesity treatment. Br J Pharmacol 2022; 179: 557-570. DOI: 10.1111/bph.15638.
11. Ussher J R, Drucker D J. Glucagon-like peptide 1 receptor agonists: cardiovascular benefits and mechanisms of action. Nat Rev Cardiol 2023; 20: 463-474. DOI: 10.1038/s41569-023-00849-3.
12. National Institute for Health and Care Excellence. GLP- 1 receptor agonists. 2024. Online information available at cks.nice.org.uk/topics/diabetes-type-2/prescribinginformation/glp-1-receptor-agonists/ (accessed December 2024).
13. Medicines and Healthcare Products Regulatory Agency (MHRA). Glucagon- Like Peptide Receptor Agonist- containing products authorised in the UK. 2024. Online information available at https://assets.publishing.service.gov.uk/media/671912baa36621334536b39d/Annex_1_GLP-1RA-containing_medicines.pdf (accessed December 2024).
14. Janić M, Škrgat S, Harlander M et al. Potential Use of GLP-1 and GIP/GLP-1 Receptor Agonists for Respiratory Disorders: Where Are We at?. Medicina 2024; 60: 2030. DOI: 10.3390/medicina60122030.
15. Haykal D, Hersant B, Cartier H, Meningaud J P. The Role of GLP-1 Agonists in Esthetic Medicine: Exploring the Impact of Semaglutide on Body Contouring and Skin Health. J Cosmet Dermatol 2024: e16716. DOI: 10.1111/jocd.16716.
16. Kong F, Wu T, Dai J, et al. Glucagon-like peptide 1 (GLP-1) receptor agonists in experimental Alzheimer’s disease models: a systematic review and meta-analysis of preclinical studies. Front Pharmacol 2023; 14: 1205207. DOI: 10.3389/fphar.2023.1205207
17. Beam W B, Hunter Guevara L R. Are serious anesthesia risks of semaglutide and other GLP-1 agonists under-recognized. Case reports of retained solid gastric contents in patients undergoing anesthesia. Anesth Patient Safety Found (APSF) Newslett 2023; 38: 69-71.
18. Fujino E, Cobb K W, Schoenherr J, Gouker L, Lund E. Anesthesia considerations for a patient on semaglutide and delayed gastric emptying. Cureus 2023;15: e42153. DOI: 10.7759/cureus.42153.
19. Gulak M A, Murphy P. Regurgitation under anesthesia in a fasted patient prescribed semaglutide for weight loss: a case report. Can J Anaesth 2023; 70: 1397–1400. DOI: 10.1007/s12630-023-02521-3.
20. Klein SR, Hobai I A. Semaglutide, delayed gastric emptying, and intraoperative pulmonary aspiration: a case report. Can J Anaesth 2023; 70: 1394–1396. DOI: 10.1007/s12630-023-02440-3.
21. Wilson P R, Bridges K H, Wilson S H. Particulate gastric contents in patients prescribed glucagon-like peptide 1 receptor agonists after appropriate perioperative fasting: a report of 2 cases. A A Pract 2023;17: e01712. DOI: 10.1213/XAA.0000000000001712
22. Raven L M, Stoita A, Feller R B, Brown C, Greenfield J R. Delayed gastric emptying with perioperative use of glucagon-like peptide-1 receptor agonists. Am J Med 2023; 136: e233-e234. DOI: 10.1016/j.amjmed.2023.07.016.
23. Weber M, Siddarthan I, Mack P F. Clinically significant emesis in a patient taking a longacting GLP-1 receptor agonist for weight loss. Br J Anaesth 2023;131: e37– e39. DOI: 10.1016/j.bja.2023.05.005.
24. Kittner S L, Talbott A L, Vishneski S R, Narbaiza J, Shields J S. Retained gastric contents after adequate fasting associated with GLP-1 receptor agonist use: a report of 3 cases. JBJS Case Connect 2023;13: e23.00506. DOI: 10.2106/JBJS.CC.23.00506.
25. Queiroz V N F, Falsarella P M, Chaves R C F, Takaoka F, Socolowski L R, Garcia R G. Risk of pulmonary aspiration during semaglutide use and anesthesia in a fasting patient: a case report with tomographic evidence. Einstein (Sao Paulo) 2023; 21: eRC0628. DOI: 10.31744/einstein_journal/2023RC0628.
27. Silveira S Q, da Silva L M, Abib A D et al. Relationship between perioperative semaglutide use and residual gastric content: a retrospective analysis of patients undergoing elective upper endoscopy. J Clin Anesth 2023; 87: 111091. DOI: 10.1016/j.jclinane.2023.111091.
28. Ye Y, Jiang B, Manne S, Moses P L et al. Epidemiology and outcomes of gastroparesis, as documented in general practice records, in the United Kingdom. Gut 2021; 70: 644-653. DOI: 10.1136/gutjnl-2020-321277.
29. Stone S, Doane M. New Antiobesity Medications, GLP-1, and GIP Receptor Agonists and their Anaesthetic Implications. 2024. Online information available at https://resources.wfsahq.org/wp-content/uploads/Tutorial_535.pdf (accessed December 2024) DOI:10.28923/atotw.535.
30. McIsaac D I, Bryson G L. Glucagon-like peptide-1 receptor agonists and aspiration risk. BMJ 2024; 387. DOI: 10.1136/bmj.q1986.
31. Green S M, Mason K P, Krauss B S. Pulmonary aspiration during procedural sedation: a comprehensive systematic review. Br J Anaesth 2017; 118: 344-354. DOI: 10.1093/bja/aex004.
32. Ruggera G, Taylor G. Pulmonary aspiration in anesthesia. West J Med 1976; 125: 411- 414.
33. British Medical Journal Best Practice. Acute aspiration. 2023. Online information available from: https://bestpractice.bmj.com/topics/en-gb/3000256#:~:text=Anaesthesia20related%20aspiration%20of%20gastric%20contents%20can%20be%20prevented,avoiding%20complete%20loss%20of%20protective%20reflexes%20from%20over-sedationhttps://bestpractice.bmj.com/topics/en-gb/3000256#:~:text=Anaesthesia20related%20aspiration%20of%20gastric%20contents%20can%20be%20prevented,avoiding%20complete%20loss%20of%20protective%20reflexes%20from%20over-sedation. (accessed 14 Feb 2024)
34. Dixon W. Use of Glucagon-Like Peptide-1 Agonists and Increased Risk of Procedural Sedation and Endotracheal Intubation in the Emergency Department. Ann Emerg Med 2024; 84: 226-227. DOI: 10.1016/j.annemergmed.2024.03.007.
35. Dixit A A, Bateman B T, Hawn M T, Odden M C, Sun E C. Preoperative GLP-1 receptor agonist use and risk of postoperative respiratory complications. JAMA 2024; 331: 1672–1673. DOI: 10.1001/jama.2024.5003.
36. Khan I. Ultrasound-guided peripheral intravenous cannulation for patients requiring dental surgery under intravenous dental sedation. Br Dent J 2022; 232: 441-448. DOI: 10.1038/s41415-022-4133-x.
37. El-Boghdadly K, Dhesi J, Fabb P, et al. Elective peri-operative management of adults taking glucagon-like peptide-1 receptor agonists, glucose-dependent insulinotropic peptide agonists and sodium-glucose cotransporter-2 inhibitors: a multidisciplinary consensus statement: A consensus statement from the Association of Anaesthetists, Association of British Clinical Diabetologists, British Obesity and Metabolic Surgery Society, Centre for Perioperative Care, Joint British Diabetes Societies for Inpatient Care, Royal College of Anaesthetists, Society for Obesity and Bariatric Anaesthesia and UK Clinical Pharmacy Association. Anaesth 2025; 80:412-424. DOI: 10.1111/anae.16541.
38. Australian and New Zealand College of Anaesthetists. Clinical Practice Recommendations regarding patients taking GLP-1 receptor agonists and dual GLP-1/GIP receptor co-agonists prior to anaesthesia or sedation for surgical and endoscopic procedures. 2025. Online information available at https://www.anzca.edu.au/getContentAsset/0f35028e-e371-4220-a49addee877051c8/80feb437-d24d-46b8-a858-4a2a28b9b970/Periprocedural-GLP-1-useconsensus-clinical-guide_P2.pdf (accessed May 2025).
39. Institute for Safe Medication Practices Canada. Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists: Risk of Aspiration during Anesthesia. 2023. Online information available at https://ismpcanada.ca/wp-content/uploads/ISMPCSB2023-i9-GLP-1.pdf (accessed December 2024).
40. Dhatariya K, Levy N, Russon K et al. Perioperative use of glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter 2 inhibitors for diabetes mellitus. Br J Anaesth 2024; 132: 639-643. DOI: 10.1016/j.bja.2023.12.015.
41. van Zuylen M L, Siegelaar S E, Plummer M P, Deane A M, Hermanides J, Hulst A H. Perioperative management of long-acting glucagon-like peptide-1 (GLP-1) receptor agonists: concerns for delayed gastric emptying and pulmonary aspiration. Br J Anaesth 2024; 132: 644-648. DOI: 10.1016/j.bja.2024.01.001.
42. Milder D A, Milder T Y, Liang S S, Kam P C A. Glucagon-like peptide-1 receptor agonists: a narrative review of clinical pharmacology and implications for peri-operative practice. Anaesth 2024; 79: 735-747. DOI: 10.1111/anae.16306.